A lot of people have asked me, “Hey Rich I have my Free SO2 results, now what do I do with it?”
Now that people are more often testing their sulfite levels in their wines, the results need to be used to the winemaker’s full advantage in protecting their wine from oxidation and spoilage. I would like to bring up the sulfite (SO2) adjustment process since the results gathered from the SC-100, SC-100A or SC-300 need to be crunched by hand or by a calculator to determine how much, if any, sulfite needs to be added to your wine.
How to adjust your sulfite levels: testing and calculating.
Whenever you move your wine from one container to another, or when you are preparing to bottle your wine, it’s important to check and adjust your sulfite (SO2) levels. What is the right level? It depends on the pH of your wine. The key parameter in protecting wine is “Molecular SO2” which for most wines following primary fermentation, should be at 0.8 ppm (mg/L). This in turn depends on the “Free SO2” (it can also be referred to as “unbound” SO2) and the pH, both of which can be measured with the Vinmetrica wine analyzers. Overall, you can reach your target Molecular SO2 by measuring and adjusting your free sulfite levels and considering your wine’s particular pH.
Measuring SO2: the Vinmetrica SC-100A and SC-300 help you to do this with confidence. But whatever methods you use, keep a few things in mind.
The key thing to check is accuracy. How do you know if your results are accurate? If you are using Aeration oxidation, you need to be sure that your NaOH titrant is standardized. A solution of sodium hydroxide loses strength over time because CO2 in the air reacts with it. Similarly, the 0.02N iodine solution that many use for Ripper titrations can undergo significant loss over time, even a relatively short period of time like 6 months. For Ripper-based methods, you can use the vitamin C trick that we recommend for Vinmetrica’s SO2 tests (although these are generally run for confidence-building, as it turns out that our reagents are very stable.)
But, you might ask, why not just make a standard solution of, say, 50 ppm SO2 from metabisulfite and analyze that to check accuracy? In principle you could do this – for example, you could weigh out fresh potassium metabisulfite (K2S2O5) accurately and dissolve it in pure water to make a 1.0% solution. This would be 10 g/L * 0.57 g SO2/g K2S2O5 = 5.7 g SO2/L = 5700 mg/L = 5700 ppm SO2. Then you could dilute this 100-fold in pure water to make a 57 ppm test solution. Here’s the problem: unless meticulous measures are taken, “pure” water is usually saturated with atmospheric oxygen, and at room temperature this level is about 8 mg/L. Each mg of oxygen will react with 4 mg of SO2, so at 8 mg/L, our water can already eat up 32 mg/L of SO2. Thus our 57 ppm (57 mg/L) SO2 solution will quickly become as low as 25 ppm. As you can see this conundrum is what brought us to use and adopt the vitamin C trick to check our SO2 reagents.
So go ahead, measure the pH and take your Free SO2 result, both can be measured with the SC-300. Make sure to write this info down! Now then, typically, if you have a higher pH wine (lower acidity), you need to add more sulfite (SO2) to achieve the Molecular SO2 level of 0.8 ppm (mg/L). The Table and graph below show the relationship between pH and Free SO2 to achieve this 0.8 ppm (mg/L) Molecular SO2. The Free SO2 on the left side of the graph indicates the Free SO2 you will NEED to achieve 0.8 mg/l Molecular SO2 in your wine. This will be made up of the Free SO2 you currently have now plus whatever you may need to add. So if your Free SO2 results (measured using Vinmetrica’s method or an alternative method) matches the Free SO2 level on the graph or table at your known pH, then you are good, no need to add more sulfite at this time. If it is not you just have to work a little harder to get it there. If your Free SO2 results are below this Free SO2 level you will need to add some sulfite to achieve this level of antimicrobial & oxidative protection for your wine. The amount you add can be determined with a sulfite calculator, which we will talk about next.
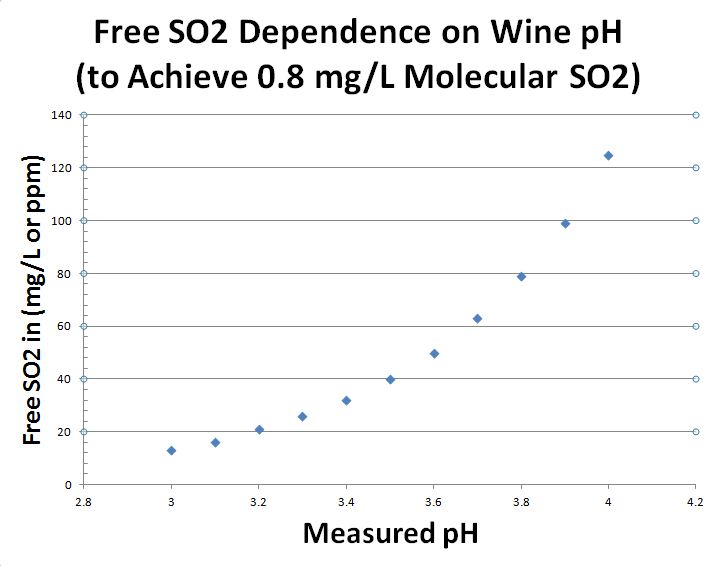
The Graph above shows the relationship of Free SO2 and pH required to achieve the 0.8 mg/L Molecular SO2. Molecular SO2 is the active form of Sulfite (SO2) which will protect your wine from oxidation and spoilage.
Table of Free SO2 concentrations necessary to attain 0.8 mg/L Molecular SO2 at a designated pH.
| pH | Free SO2 ppm (mg/L) |
|---|---|
| 3.0 | 13 |
| 3.1 | 16 |
| 3.2 | 21 |
| 3.3 | 26 |
| 3.4 | 32 |
| 3.5 | 40 |
| 3.6 | 50 |
| 3.7 | 63 |
| 3.8 | 79 |
| 3.9 | 99 |
| 4.0 | 125 |
Wait I have to do this by hand? No fear not, that is what the sulfite calculator is for! Winemaker Magazine’s website provides a great calculator and except for some tweaks, which we will talk about below, the instructions are pretty clear.
Once you know your sulfite levels, it’s easy to make any adjustments that may be necessary. The table and graph above is there to guide you in this, but we always like to use the sulfite calculators that are available for download or on the web as applications. My favorite is the Winemaker Magazine Sulfite calculator on the web:
WineMaker Magazine Sulfite Calculator
This is easy to use and is accompanied by a nice guide. However a few notes are in order:

1. I highly recommend you choose (and use) “10% Sulfite Solution” from the drop-down menu next to the prompt “Preferred method of Sulfite addition”. (The guide tells you how to prepare the 10% solution. The sulfite solution we’re talking about is potassium metabisulfite (KMBS), though if you choose to use sodium metabisulfite or other sulfite forms, the accuracy is not too bad.).
2. Choose the wine type (red or white). Keep the desired molecular SO2 at 0.8 and enter your wine’s pH value (hit your keyboard’s “Enter” button after entering the value).
3. This is the one step that is not obvious at first (at least it wasn’t to me). After you enter your wine’s pH and the molecular SO2 level, you will see a recommended free SO2 level in the “Notes:” section, just below the entry fields. Enter this number in the “Desired level of free SO2” field. If you don’t see anything in the “Notes” section, enter a “guess” number higher than your current level in the “Desired level of free SO2” field (and see step 6!).
4. Next you enter the “Current level of free SO2” (What you have just measured), and the “Volume of wine to be corrected”, being sure to indicate liters or gallons as appropriate.
5. Now by pressing the “Calculate” button below, you should see a figure under “Amount of sulfite to be added in milliliters (if you used Liters for the wine volume) or fluid ounces (if you used Gallons).
6. If you entered a “guess” number in step 3, you now should see the recommended level in the Notes: section – so enter this and repeat the calculation.
Simple as that! Now that you have protected your wine from oxidation and spoilage, seal up your container and take a siesta. But remember to check your wine for sulfite regularly, especially after you transfer your wine to a new container or are about to bottle!

I just tested my SO2 on my latest batch of blueberry wine. The PH was 3.18 and the SO2 was for all intensive purposes, 100 ppm, (I used pretty much the entire 5 ml syringe to get my 20 beeps). It seems with a PH of 3.18, I only needed an SO2 level of around 21 ppm. The wine was started only 2 weeks ago and already my hydrometer says I have 0.990, completely dry. So I plan to let the wine age in carboys under air locks for at least 2 or three months before I rack again. Is it likely the SO2 will come down a little closer to what is needed to protect the wine and if it doesn’t, do I need to try to figure out a way of lowering my SO2 level? TA was at 6.8
John,
first let’s be sure that your SO2 numbers are good. Some versions of our instruments have intermittent problems with correct signaling of the endpoint (it suddenly comes to life, but way after the endpoint has been passed, leading to erroneously high results). If your instrument has an RCA (phono-style) connector for the SO2 probe, it may need physical adjustment to ensure good contact of both conductors (contact us for more information). The older and newer versions have BNC (twist-lock) connectors that don’t have this problem. Try the test for sensitivity “3. Connect the electrode and put it in about 20 mL of distilled water; add about 1 ml (half a bulb squeeze) of each of the acid solution and the reactant and swirl in the usual way keeping constant motion. The instrument may or may not indicate STOP as above. If it does not, add a drop of the SO2 Titrant solution. This should make the STOP condition occur. [If it doesn’t you may have an electrode problem read in the next section below how to fix this.] Now add one drop of a concentrated sulfite solution (1-10% is fine) and verify that the STOP signal ends and the PROCEED light illuminates. If this happens, your electrode is probably OK as well.”
If indeed you have free SO2 around 100 ppm, it probably won’t drop much in a glass carboy because those are pretty good at keeping oxygen out. But in this case I would suggest that you not do anything because 100 ppm, though higher than you need it to be, will probably not affect the taste sensation too much, and it is not a health risk. In other words, let your palate be the guide. If you sense the off aroma of burnt matches due to excess SO2, you can add something like hydrogen peroxide (a few drops of the 3% stuff from the store per carboy at a time) or just splash the wine in air to reduce the SO2 levels.
Regards,
Rich
What is a sulphite- eater?I struggle to stablelise my Merlot 2013 with So2.I have put so2 twice in this week.I want the wine to be stabilized at least at 30ppm before botteling it. Two days after putting in 40ppm the FSO2 is less than 10 ppm.The TSO2 is at about 80ppm on this stage
Jan, there are several components of wine that can bind SO2, like sugars, phenolic compounds, anthocyanins, and other (see for example, http://www.practicalwinery.com/janfeb09/page2.htm ). Also see our FAQ under “I added the right amount of sulfite to my wine but the numbers are still low!”:
This is a common occurrence with several explanations, any or all of which may be happening:
Make sure you are using fresh sulfite powder. Potassium metabisulfite degrades over time and that stuff you bought 2 years ago is probably bad now!
Make sure that you stir your wine thoroughly when you add sulfite. If you pour a 10% solution of KMBS into your wine, it sinks like a battleship! A sample taken off the top will read low unless the wine is stirred.
A significant portion of the sulfite you added may have ended up ‘bound’, particularly if your free SO2 was very low to begin with. This bound SO2 does not show up when you measure free SO2, and it is not protecting your wine. You will need to add more sulfite until your free SO2 comes up to the right level. Sometimes you must add 2 or even 3 times more sulfite than you first calculated
Regards,
J.Richard Sportsman
i got the 300 meter and I do the test for so2 just as your paper work says, When I added the sulfite the wine foams up. Do you know why it does that? I also notice that once I do that it keeps bubbling up for days, is that normal?
I got 8 carboys (5gal) I never checked because I didn’t have a meter, is it proper to check even if I don’t need to rack yet? If they been sitting for 6 months or more still, is it normail for them after I sulfite to start again?
Phil,
Foaming is a bit peculiar when adding potassium metabisulfite. I would make sure that the sulfite level was increased after adding potassium metabisulfite (KMBS) to your wine. If not, is it possible you added something like potassium carbonate by mistake? That would explain the foaming and the bubbling for a number of days. I will admit I have mixed up bags of crystalline solids before, so I would monitor your pH levels and your SO2 levels in your wine to compare to where you were before adding the sulfite. I recommend making a 10% W/V KMBS solution in distilled water. For example take 100 g of KMBS and put it into a 1-Liter glass container, then add distilled water until you reach a final volume of 1 L. Mix the potassium metabisulfite solution well, until the KMBS is completely dissolved, then calculate the volume necessary to add to your wine using a sulfite calculator like this one.
I would go ahead and check your sulfite levels every couple of months if you are unsure. While carboys typically don’t have the same oxidation and evaporation qualities of an oak barrel, the sulfite levels do change over time. Luckily our tests don’t take too much time and are relatively inexpensive, so measure as frequently as necessary. Have a Happy New Years.
Regards,
Rich Jr.